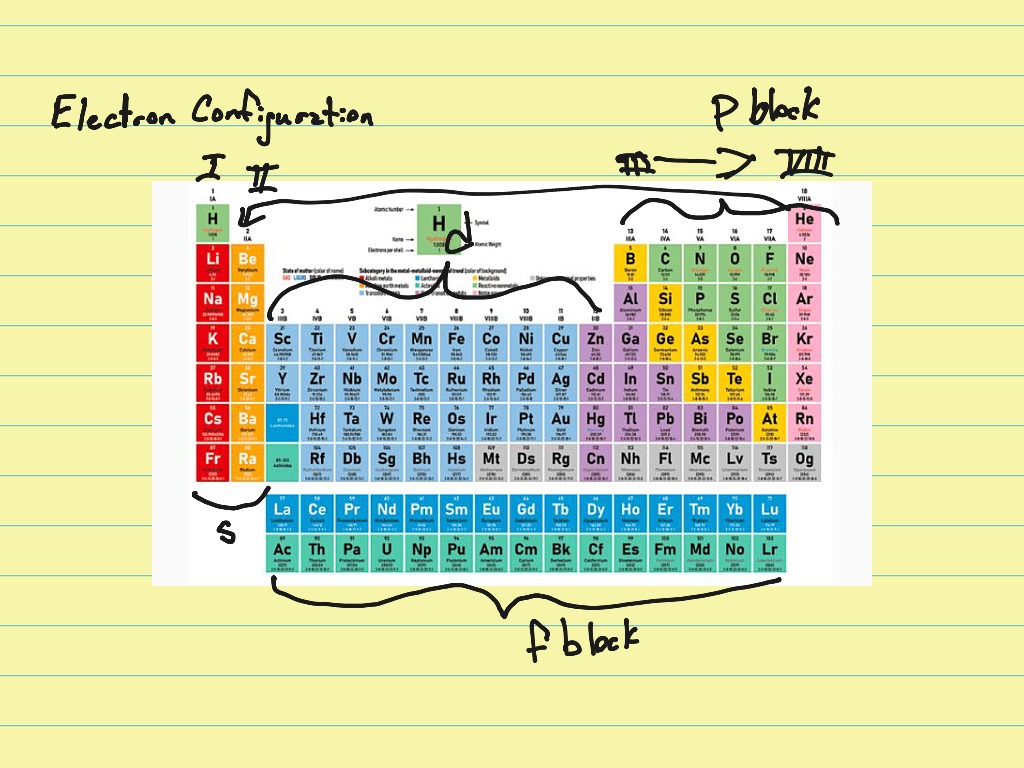
This electronic configuration can also be shown as a diagram. This electronic configuration can be written as 2.8.1 (each dot separates one shell from the next). Sodium atoms have 11 protons and so 11 electrons: It lists the orbital symbols sequentially. For example, the atomic number of sodium is 11. As previously mentioned, electron configuration is a particular distribution of electrons among available orbitals. The electronic configuration of an atom can be predicted from its atomic number. For elements with atomic number 1 to 20: Electron shell They begin to occupy the next shell when a shell becomes full. Electrons occupy shells starting with the innermost one. Electrons in shellsĭifferent shells can hold different maximum numbers of electrons. The S orbital is spherical, the P orbital is dumbell shaped, D orbitals are shaped like four-leaf clovers and F orbitals are a lot more complicated as seen in the image.An electronic configuration is the way in which electrons are arranged in an atom. The general electronic configuration of group-14 elements is ns2 np2. There are 4 different orbital shapes, S, P, D and F. Carbon, silicon, germanium, tin, and lead in the periodic table belong to group-14 or IVA. When treated as waves they can be assigned different properties. But electrons can also be treated as waves. 
Electrons as previously explored are treated as particles, negative particles with a low mass that orbits the nucleus. To understand this we need to explore the concepts of electrons when considered as quantum particles. Extending understanding beyond shells it can be explored that there are subshells. The electron configuration of elements plays an even deeper role in the structure of the periodic table. This gives you the electron configuration in the correct order! You can progressively write S subshells, P subshells, D subshells, and F subshells in columns, skipping an energy level in each progressive column and draw diagonal lines through the columns from the top right to the bottom left. How do you do the electron configuration shortcut? The number of electrons in a shell can be determined by the general formula 2n2, where n is the shell number (energy level).ĥ. See below for a helpful way to remember this! The electron configuration describes the position of electrons of an atom or a molecule in atomic or molecular orbitals. (a) If each core electron (that is, the 1 s electrons) were totally effective in shielding the valence electrons (that is, the 2 s and 2 p electrons) from the nucleus and the valence electrons did not shield one another, what would be the shielding constant ( ) and the. Starting from the innermost subshell to the outermost, the progression of electron subshells is as follows: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p, 8s. The electron configuration of B is 1 s 2 2 s 2 2 p 1. What is the electron configuration order? Electrons in the outermost orbitals, called valence electrons, are responsible for most of the chemical behavior of elements. First start by determining the electron configuration, then write the subshells with how many electrons they possess from closest to the nucleus to the furthest.ģ. The electron configuration is the list of subshells where the electrons in an atom are located. How do I write an electron configuration? See the examples below for a detailed explanation.Ģ. In quantum chemistry and atomic physics, the distribution of electrons of a molecule or an atom in the molecular or atomic orbitals is known as electronic. How do I determine the electron configuration?Ĭheck out the periodic table to find out how many electrons an atom has, draw out the progression of subshells, and fill the orbitals of the energy levels one by one until all electrons have a place. An element’s group number on the periodic table is equivalent to the number of electrons in its outermost shell.ġ. The different shells can hold different numbers of electrons. Each shell can only contain a given number of electrons. Refresher: Electrons exist in shells, orbiting around an element’s nucleus. electron configuration Electrons are arranged around the nucleus in shells of well defined energy. One cool detail that you might notice is that the electron configurations of successive elements (ordered by their periodic number) contain each other.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
